An unforgettable award from Anna Falanga & Alok Khorana
An unforgettable award from Anna Falanga & Alok Khorana
Our SERENITY Consortium is proud of Elin Baddeley’s achievement at 13th ICTHIC – International Conference on Thrombosis and Hemostasis Issues in Cancer in Bergamo!

CoClarity trial
CoClarity trial
We are proud to share an update on SERENITY. As part of this project, the CoClarity trial is evaluating the appropriate use of antithrombotic therapy near the end of life.
The CoClarity trial explores how a Shared Decision Support Tool can help patients and clinicians make better informed decisions together.
The study is currently running in the Netherlands, Spain, Poland, and the United Kingdom. First patient inclusions have started and the collaboration continues to grow.
This marks an important step toward more person centered and appropriate care.

Special webinar on the SERENITY Study
Special webinar on the SERENITY Study
We are excited to invite you to a special webinar on the SERENITY Study and its potential to change clinical practice in cardio-oncology.
We would like you to join us on 9 April at 14.00 GMT (15.00 CET) for this Webinar.
In this webinar, the following guest speakers from our consortium will present:
- Dr Michelle Edwards – Cardiff University, UK
- Dr Anette Arbjerg Højen – Danish Center for Health Services Research, Denmark
- Dr Stella Trompet – Leiden University Medical Center, The Netherlands
*Seminar Alert!* Registration is now open for our February Seminar!
*Seminar Alert!* Registration is now open for our February Seminar!
Topic: “SERENITY: developing an evidence-based decision support tool to support shared decisions about continuing or discontinuing antithrombotic medication for people with cancer towards the end-of-life” Presented by Dr. Michelle Edwards & Dr. Kathy Seddon
When: Friday 27th February 2026 [UTC 1:30 PM | CET 2:30 PM | BST 1:30 PM | EST 8:30 AM | CST 7:30 AM] Check your local time
Cost: Free
*A recording of the seminar will be made available exclusively to ISDMS members on the Society’s website approx. 1 week after the session.

We are developing a decision support tool
We are developing a decision support tool
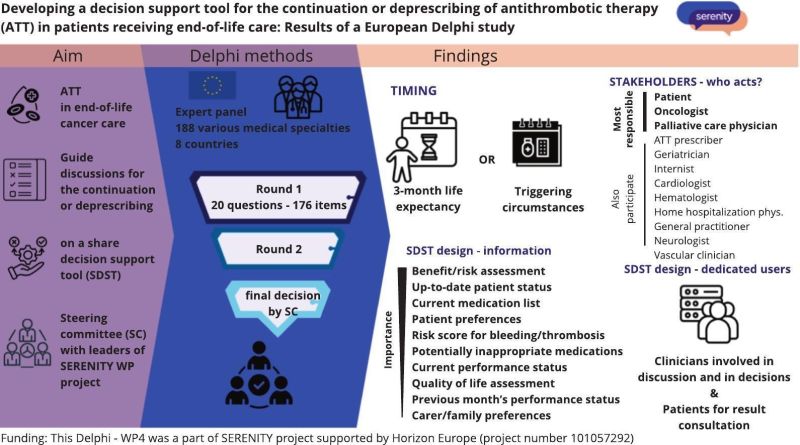
We are developing a decision support tool for the continuation or deprescribing of antithrombotic therapy in patients receiving end-of-life care: Results of a European Delphi study.
Serenity joins the Quality of Life and Patient-Centred Care (QoL-PCC) cluster
Serenity joins the Quality of Life and Patient-Centred Care (QoL-PCC) cluster
The cluster represents a key opportunity to advance oncology care. One of the main goals is to develop a unified vision around these concepts while establishing core axes related to Patient and Public Involvement (PPI) and stakeholder engagement.
Promoted by the European Commission and led by EUonQol, the cluster brings together projects such as PREFERABLE-II, EU Navigate, MyPath_EU, Pal-Cycles, INSPIRE Palliative Rehabilitation Project, RELEVIUM Project and SERENITY, all EU-funded projects working to advance palliative oncology care.
WP4: Delphi Process
WP4: Delphi Process
Through this Delphi study, the following aspects will be defined:
- characterisation of candidate patients for discussion about ATT deprescribing
- healthcare team roles in ATT decision-making
- specific information and communication requirements for patients when making deprescribing decisions
- SDST content priorities; and optimal outcomes for the planned clinical trial.
Who am I to stop taking my medication?
Who am I to stop taking my medication?
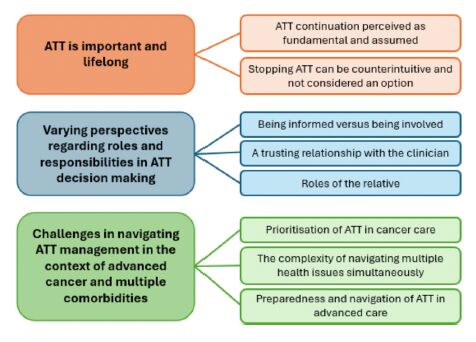
Who am I to stop taking my medication – 𝘢 𝘱𝘢𝘵𝘪𝘦𝘯𝘵 𝘢𝘴𝘬𝘦𝘥 𝘪𝘯 𝘰𝘶𝘳 𝘳𝘦𝘤𝘦𝘯𝘵 𝘴𝘵𝘶𝘥𝘺.
When we as professionals want to stop medication, we often focus on the clinical reasoning: Risk of bleeding. Limited benefit. A burden.
All true — but not enough.
Patients need more than facts. They need support to engage in that decision. And some simply don’t feel it’s their place.
This is one of the key findings in our new qualitative study, part of the SERENITY consortium:
Some patients are willing and able to co-decide about stopping antithrombotics
Others defer to clinicians, feeling it’s not their role — especially near the end of life
The difference isn’t about education or attitude. It’s about context, trust, timing, framing.
Advance care planning isn’t just about what we do — but how we decide it. Together. Stopping medication starts with the patient.
New publication in British Journal of Haematology
New publication in British Journal of Haematology
Retrospective cohort study of adults in Wales diagnosed with poor prognosis cancer between 2013 and 2021, following up patients from cancer diagnosis until death, end of follow-up or study end (31 December 2021). Outcomes included ATT discontinuation, bleeding and thromboembolic events in secondary care.
New publication about Shared decision-making
New publication about Shared decision-making
This realist synthesis explores shared decision-making (SDM) to:
(a) provide insights into why prescribing continues in end-of-life care;
(b) build a conceptual platform for optimizing ATT prescribing for persons living with cancer towards end-of-life.
Conclusion
Implementation of ATT deprescribing is enabled or constrained by
(a) the meaning of medications to patients;
(b) clinician engagement and understanding;
(c) multi-disciplinary clinical decision-making processes (including support tools);
(d) patient empowerment;
(e) organizational investment.